- Research paper
- Open access
- CCA4.0 Intern’l License
- Not for the profit
Evans Afriyie-Gyawu,1 Padmini Shankar,2 Gulzar H. Shah, 3 Alicia Brown,3 Shannon Cearley,4 Akwasi Ampofo-Yeboah5 Nathaniel Abrams,1 Jian Zhang,1
Metadata
DOI: https://doi.org/10.61915/pnd.120400
Abstract
Introduction
Lycopene is a carotenoid found naturally in some fruits (such as watermelon, papaya, and pink guava) and vegetables (such as tomatoes, sweet bell peppers, and asparagus) (1). Lycopene is the red pigment in tomatoes and tomato products, and it has been found that increasing dietary intake and blood concentration reduces the risk of prostate cancer and cardiovascular disease (2).
Tomatoes are the most abundant source of dietary lycopene, accounting for 85 percent of total dietary lycopene, with nutritional values ranging from 0.9 to 9.27mg/100g (3). Lycopene levels are higher in concentrated foods such as ketchup, tomato paste, and tomato sauce than in unprocessed tomatoes (4).
It is important to note that heating (while cooking) causes cell wall breakdown, converting the natural all-trans form of lycopene to its cis-isomerized state and increasing its bioavailability in the intestine (5) However, lycopene bioavailability from dietary sources is low, and long-term consumption of high levels of dietary calcium (found in dairy products) and some mineral water products can further reduce lycopene absorption, partly due to its hydrophobic nature (2).
Lycopene bioavailability is estimated to be 5% when consumed as tomato juice and 23% when consumed as pure lycopene mixed in olive oil. Another reason is that calcium binds to some food components, which reduces bioavailability when eaten with foods containing lycopene due to “partial/total masking of its inhibitory effect on lycopene micellization” (2).
In vitro studies have shown that “calcium cations bind to the negatively charged micelles and thus diminish their charge,” influencing lycopene uptake by enterocytes. Even when combined with 500 mg of calcium (half of the RDA for men), lycopene’s bioavailability is reduced (2). Another factor to consider is the half-life of lycopene in the human body to determine the frequency of consumption to maintain consistent plasma levels (6).
Lycopene, Lung Cancer and Other Cancers
According to a report on the potential relationship between some cancers and lycopene, lycopene may play a role in reducing the progression of lung and prostate cancers (7). Although little is known about the mechanism, it may be similar to its protective mechanism against disease development (8).
Because of their antioxidant properties, fruits and vegetables should be consumed daily to reduce the risk of developing certain cancers (5, 9). Antioxidants protect vital molecules in the body from oxidation by removing free radicals (10, 11, 12). Lycopene is a powerful antioxidant, and several studies indicate that it may reduce the risk of developing certain cancers such as lung, prostate, and breast cancer (13).
Several epidemiological studies have found a link between a lower risk of lung cancer and other cancers in populations that consume tomato-rich diets high in lycopene (14, 15). Even with these findings, the precise role of lycopene in the prevention of lung cancer cell growth remains unknown, necessitating further controlled clinical trials (1313).
Some mechanisms that have been proposed to explain how lycopene can suppress carcinogenesis include the use of its antioxidant properties, the induction of cytoprotective ‘phase II’ enzymes, decreased lipid peroxidation, and the inhibition of cancer cell proliferation (13).
Behavioral and other risk factors have also been linked to the development of lung cancer and death from it. Cigarette smoking is the leading cause of lung cancer, accounting for approximately 80% to 90% of lung cancer deaths in the United States (16). Tobacco use causes oxidative stress in lung cells, and the presence of free radicals in cigarette smoke causes oxidative stress, disrupting the balance between oxidation and reduction (17).
Over 4,000 chemicals are found in cigarette smoke, including “nicotine, carbon monoxide, acrolein, and oxidant compounds” (18). Furthermore, smoking causes inflammation, which suppresses the immune system, allowing cancer cells to avoid detection, progress, and damage the DNA. Inhalation of dust, ozone, mineral fibers, air pollution, and tobacco smoke have been shown to cause inflammation within the epithelial tissue of the lungs, which has been linked to the onset of respiratory diseases such as lung cancer (18).
Lycopene, a well-known antioxidant, has the highest potential antioxidant effect of all carotenoids (13), and its consumption has been linked to a lower risk of lung cancer (9). Lycopene and beta-carotene were found in lung tissues in a post-mortem study. This lends credence to the notion that carotenoids are a component of the lung’s defense mechanism against cancer formation (19).
Even though “tomatoes and tomato products containing lycopene have been associated with a lower risk of malignancies, particularly prostate cancer,” “many dietary-based epidemiological studies have yielded less supportive results” (5). Furthermore, while randomized controlled trials in prostate cancer studies found no effect of lycopene, meta-analyses of limited observational evidence found that only high lycopene intake has potential effects (20).
Lycopene has been shown in laboratory studies to inhibit cancer cell growth or prevent carcinogenesis when chemically induced (13). Although experimental evidence is promising, clinical evidence to support the use of lycopene supplements for cancer prevention is currently lacking (13, 21).
This may be due, in part, to evidence from recent studies indicating that lycopene is more effective in preventing certain cancers when consumed in tomato form rather than as a purified supplement (22). In a double-blind randomized controlled trial, for example, high doses of lycopene and other nutrients were shown to have no significant biomarker levels of prostate-specific antigens (PSA) between the treated and control groups. In fact, at the end of the supplementation period, re-biopsy revealed that the treated group had a higher incidence of prostate cancer (21).
While numerous studies have shown a link between lycopene and protection against the initiation and/or progression of lung cancer, there is limited data and research on its effect on lung cancer survivability (19).
Objective
Thus, this study aimed to determine the relationship (or lack thereof) between serum levels of lycopene and lung cancer mortality. We also aimed at examining whether dietary factors are associated with serum levels of lycopene.
Materials and Methods
Sample Size and Characteristics
This lung cancer study used data from the National Health and Nutrition Examination Survey III (NHANES III), which was conducted between 1988 and 1994. The National Health and Nutrition Examination Survey (NHANES) is a major program of the National Center for Health Statistics (NCHS), which is part of the US Centers for Disease Control and Prevention (CDC) and is responsible for producing vital health statistics for the United States.
Every year, the NHANES examines a representative sample (from the US population) of approximately 5,000 people (23). NCHS used a two-phased multistage probability sampling of the noninstitutionalized United States population. Phase I lasted from October 18, 1988, to October 24, 1991, and Phase II lasted from September 20, 1991, to October 15, 1994.
This study’s dataset was linked to the National Death Index (NDI) database, which included American adults over the age of seventeen who were followed up to December 2006. The NCHS connects survey participants’ data with NDI mortality data, allowing for a variety of outcome studies to be conducted to investigate the relationship of a wide range of health factors with mortality (24).
To determine the vital status and causes of death among the study participants, a total of 12 identifiers (including social security number, gender, and date of birth) were used to link NHANES III participants with the NDI (25).
Only people aged 19 and up were eligible to participate from this pool. They were not included in the study population if there was no data on their family income, drinking, smoking, educational level, or vegetable consumption. Following the application of all inclusion and exclusion criteria, a total of 14,358 final participants (as shown in Table 1) were evaluated for the study.
The NHANES protocol was reviewed and approved by the National Center for Health Statistics’ Institutional Review Board (IRB). Because only a public dataset was used, the current study was exempt from ethics review by the authors’ university’s IRB
This dataset in Table 1, was acquired during in-home interviews and subsequent visits by the study participants to a mobile examination center as part of the NHANES III. Standardized questionnaires were used to collect the demographics and health-related information used in the study.
Socioeconomic Status (SES)
The poverty income ratio (PIR) was calculated using the previous year’s income and family size to determine the family income level. A PIR1 is defined by the federal government as being below the poverty line. Income was divided into four categories for our study: high-income [PIR=4+], middle-income [PIR=2-3.99], low-income [PIR=1.00-1.99], and poor [PIR1].
Educational attainment was defined as the highest level of education completed and was classified as college graduate and above, some college, high school, or below. Marital status was classified as either married or single. The race and ethnicity category was reduced to four options: African American, White American, Mexican American, or Other.
Health Risk Factors
Smoking was defined as “heavy” if respondents reported smoking ≥40 cigarettes in the past 5 days, and as “moderate” if <40 but >0 cigarettes were smoked in the past 5 days. Self-reported health was measured as excellent, good to very good, or poor.
Classification of body size was determined using BMI generated from the weight in kilograms divided by the square of their measured height in meters. Body size was categorized as “underweight” (BMI<18.5), “normal” (BMI of 18.5-24.9), “overweight” (BMI of 25.0-29.9), and “obese” (BMI ≥30). Vegetable and fruit/juice consumption were measured as <1 per day, 1-2 per day, or 3 or more per day. Cancer status was established at baseline and was determined to be a “positive” or “negative” diagnosis from a physician at the time of the first assessment.
Analysis of Serum Lycopene
Trained technicians used a standardized protocol to collect blood samples from study participants while maintaining controlled and consistent environmental conditions. These specimens were examined in a mobile examination center. Whole blood was collected into serum separator tubes without anticoagulant treatment and held at room temperature for 30–60 minutes before centrifugation. Serum was separated, frozen at 20°C, and transported on dry ice to the Centers for
Disease Control and Prevention (CDC) central laboratory for priority analyses. The remaining samples were then stored at -70oC for 8 months to 5 years pending lycopene and other micronutrient concentration analyses during Phase II of the NHANES III. Based on the detected serum lycopene levels, a distribution curve was created. Lycopene had a detection limit of 0.63 g/dl. Sex-specific cutoffs were derived from lycopene levels in patients’ sera to divide the study participants into three groups:
The distribution’s upper quartile (i.e., high group) included males with lycopene serum levels of 33.51 g/dL and females with 29.67 g/dL. Males with 18.14 g/dL and females with 16.47 g/dL were in the lower quartile (i.e., low group hereafter). The middle quartile (i.e., moderate group) of lycopene levels was found between the upper and lower quartiles for both male and female participants. With regard to fatalities and other clinical data, all laboratory measurements were performed blindly.
Fatality Follow-up and Identification of Causes of Death
A total of twelve identifiers (including social security number, gender, date of birth, and other demographics) were used to connect NHANES III participants to the National Death Index to obtain vital status and cause of death.
Over 96% of the deceased participants and nearly all of the living participants were successfully tracked down and classified. The person-year (PY) contributions of each participant were calculated as the time between the baseline examination and the person’s death or December 31, 2006. (if alive).
The average length of follow-up was 10.46 years, with a maximum of 18.08 years. The cause of death was determined using the underlying cause listed on the death certificate. To code the cause of death, which in this study was lung cancer, the International Classification of Diseases, Injuries, and Causes of Death (ICD) (10th Revision) was used. To standardize the codes, the NCHS recoded all deaths that occurred before 1999 that were coded using the ICD-9 system with the comparable ICD10 code.
Statistical Analysis
Software for Survey Design Analysis (SUDAAN) was used to analyze the data. Statistical tests were determined to be statistically significant at a p-value of 0.05. Hazard Ratios (HRs) were estimated using the Cox Proportional Hazards Regression. The Statistical Analysis System (Version 9.4) software was used to obtain unweighted estimates.
The survival times were estimated with the Kaplan–Meier method. The estimates of the survival probability function were obtained from Cox proportional hazard regression models for individuals with different lycopene levels. The death rate (i.e., the number of deaths per 1000 person*years), was calculated separately for the low, moderate, and high lycopene groups.
Cox Proportional Hazard Regression models were used to estimate the crude and adjusted HRs of dying from lung cancer with varying concentrations of serum lycopene levels. HRs were also adjusted for age, sex, and other possible confounders like vegetable consumption, race, level of education, and family income.
Results
The majority (42.19%) of the participants had moderate serum levels of lycopene followed by low levels (38.88%) and then high levels (18.93%). . Among individuals who had low levels of serum lycopene at baseline, about 48.91% had cancer, and 38.07% did not have cancer. This trend was reversed among individuals with moderate (37.35% – cancer and 42.58% – no cancer) and high (13.74% – cancer and 19.35% – no cancer) categories of serum lycopene levels at baseline.
The total deaths from all causes of cancer among individuals aged 60+ in this study were calculated to be 4.95%. Of this amount, lung cancer mortality accounted for 29.63%. These data suggest an inverse association between serum lycopene levels and lung cancer mortality.
Table 2 illustrates the association between specific population characteristics and serum levels of lycopene. African Americans (20.84%) and White Americans (19.44%) were significantly more likely to have high serum levels of lycopene compared to Mexican Americans (16.01%) and “Others” (18.80%). Generally, those with lower education, income, and self-reported health levels had significantly lower serum levels of lycopene.
There were significantly more individuals who reported their health as “poor” (51%) that had low serum levels of lycopene compared to those who reported “good/very good” (39.35%) or “excellent” (31.77%) health. This trend reversed as the serum lycopene levels increased from moderate to high.
The educational level of most of the study participants was high school or less. There was a significant relationship between low levels of serum lycopene and low levels of educational attainment.
For instance, participants whose highest level of education was high school (42.47%) were significantly more likely to have low serum levels of lycopene compared to people who completed some college (29.79%) or were college graduates (34.05%). There were noteworthy bi-variate associations between lycopene and deaths due to cancer in adults older than sixty years.
As illustrated in Table 3, about 58.85% of all-cancer mortalities were found in individuals who had low serum levels of lycopene compared to those who had moderate (29.92%) and high serum lycopene levels (11.24%).
With lung cancer mortalities, there was also a significant association with serum levels of lycopene. Approximately 61.61% of lung cancer mortalities were found in individuals who had low serum levels of lycopene compared to those who had moderate (27.49%) and high serum lycopene levels (10.90%).
Table 3: HRs were used as a form of additional analysis of the cancer mortality data. Unadjusted and adjusted HRs analyzed the association of low serum levels (which is less than 25% of all the serum lycopene concentrations) of lycopene with cancer-related deaths (of which lung cancer was inclusive). This was then adjusted for age, sex, race, family income, educational attainment, alcohol intake, and smoking behavior.
Table 4: The HR for low serum levels of lycopene and lung cancer mortality were found to be significantly associated, with an HR of 1.67 (1.03-2.71) and a death rate of 1.99 as illustrated in Table 4. Results also show a link between an increased risk of lung cancer deaths and low serum lycopene levels. There was also a significant association between low serum levels of lycopene and both the unadjusted and adjusted HR (for age, sex, race, family income, education attainment, alcohol drinking, cigarette smoking, fruits, and vegetable consumption).
Table 5 shows that after adjusting for age, sex, race, family income, education, alcohol drinking, and cigarette smoking, study participants with low serum lycopene levels had 1.45 (1.08-1.96) times increased risk of lung cancer mortality, and 1.27 (1.08-1.49) times greater risk for all-cancer deaths compared to the high serum lycopene levels (ref=1). Unadjusted low serum lycopene levels can also increase the risk for lung cancer mortality by 1.67 times (1.03-2.71).
Using a three-level serum concentration categorization (i.e., low vs. high and moderate vs. high), low levels of lycopene were significantly associated with 1.67 (1.03-2.71) increased risk of lung cancer mortality compared to individuals with high serum lycopene levels even after adjusting for fruits and vegetables.
Table 6 shows that the unadjusted HR for fruits and vegetables was significant at 1.68 (1.04-2.72), thus indicating that there was a 68% higher risk of death from lung cancer in people who had low serum levels of lycopene when compared to study participants with high serum lycopene concentrations. Similar findings are tabulated for the HR adjusted for fruits and vegetables, with an HR of 1.67 (1.03-2.71). Also, the unadjusted HR for fruits and vegetables was significant at 1.30 (1.01-1.68), thus indicating that there was a 30% higher risk of death from all-cancer in people who had low serum levels of lycopene compared to study participants with high serum lycopene levels. While the adjusted HR for fruits and vegetables appears to be 28% higher risk of death from all-cancers (HR=1.28) among individuals with high serum levels compared to low levels, the associated risk is not statistically significant (CI, 0.99-1.65).
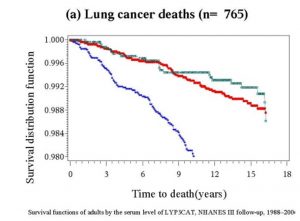
The survival functions displayed in Fig 1 provide a visual representation of the association between serum lycopene levels and lung cancer survival. In the graphs, blue, red, and green lines represent low, moderate, and high serum levels of lycopene, respectively. Decreased survivability correlated with low serum levels of lycopene. The most pronounced difference was seen in those with low serum levels having dramatically lower survivability compared to moderate or high serum levels
Discussion
Because of its protective role in cancer prevention and progression, the link between serum lycopene levels and cancer mortality warrants further investigation. We discovered that lower serum lycopene levels increased the risk of death from lung cancer in our analysis of the NHANES Survey (1988-1994), which is a national representation of the US population. This lung cancer study discovered a link between lung cancer mortality and lycopene levels in the blood.
Lycopene is an antioxidant that can be found in both natural and processed foods, such as tomato peels (5, 9, 13). As previously stated, several studies have been conducted to investigate lycopene serum levels and their role in cancer prevention and progression (7, 11, 13, 14, 15). According to the current lung cancer study, the majority of the study population had moderate serum levels of lycopene, and those with cancer were more likely to have lower levels of lycopene than those without cancer.
Although some studies have found no link between lycopene consumption and the risk of certain cancers (26), our findings are consistent with previous research that found an inverse relationship between lycopene levels in the blood and the development of lung cancer. This could be due to lycopene’s role in preventing oxidation, which can lead to gene mutation and carcinogenesis (27).
Another finding from this lung cancer study was that a slightly higher percentage of heavy smokers (41.90 percent) had low levels of lycopene than those who never/rarely smoked (38.18). Furthermore, a previous study found that confounders such as smoking lower plasma carotenoid (e.g., lycopene) levels in smokers compared to non-smokers (28).
This could be due to several factors, including the fact that smoking inhibits lycopene function and is a major risk factor for the development of lung cancer (29, 30). It is unclear whether smoking status influences serum lycopene levels in humans in this study. These similarities support the association found in our study between low serum lycopene levels in smokers and lung cancer patients.
Our findings suggest that low lycopene levels in the blood are linked to an increase in lung cancer deaths. According to the survival functions chart, patients with low lycopene serum levels had lower survivability than those with moderate or high serum levels.
This could be related to the fact that lycopene slows the progression of lung cancer in humans by inhibiting the p53 gene, which is required for cancer development (31). Thus, increasing dietary lycopene intake or lycopene supplementation may play a role in lowering lung cancer mortality.
One of the study’s strengths is the possibility of generalizability of our findings. Using the NHANES as our data source ensures that the study participants are representative of the national population rather than just individuals in clinical settings. As a result, the contribution to the body of knowledge surrounding lycopene could have a positive impact on people’s lives and enhance sustainable development in the United States and around the world.
The NHANES sample size is sufficient for a thorough analysis of the data. Furthermore, to the best of our knowledge, this is the first study to look at the relationship between serum lycopene levels and lung cancer mortality in the American population. The majority of the research found focused on serum lycopene and cancer prevention. This lung cancer study, on the other hand, looked at lycopene levels in the blood and their relationship to lung cancer mortality.
We are, however, aware of the study’s limitations. Serum lycopene levels were only measured once (at baseline) during this study, so it does not account for the effect of participants’ habitual or sporadic intake of fruits and vegetables, which could affect serum lycopene levels. Individuals in institutionalized facilities were not included in the study population for this research.
These included the elderly in hospices and nursing homes, as well as young people in correctional facilities. The NHANES data also excluded sensitive population groups such as pregnant women and young children.
There is also the possibility of misclassification of causes of death on death certificates, as individuals with lung cancer may die from causes other than the disease itself. Because the NHANES dataset only provides a cross-sectional study and analyses, an effort must be made to conduct real-time follow-up with the population to assess the applicable role of serum lycopene in chronic diseases, specifically lung cancer mortality.
Despite these limitations and the fact that the data are not very recent, this lung cancer study contributes significantly to filling gaps in existing research on serum lycopene levels and the risk of lung cancer mortality.
Conclusion
This study provides evidence that there is a link between lung cancer mortality and lycopene levels in the blood. According to the findings of the lung cancer study, there is a significant difference in lung cancer mortality between individuals with low serum lycopene levels and those with high serum lycopene levels. As a result, it is possible that lycopene could reduce the risk of lung cancer development and possibly improve the chances of survival in lung cancer patients.
Previous research suggests that confounders such as smoking can lower plasma carotenoid (e.g., lycopene) levels in smokers compared to nonsmokers. Understandably, the relationship between lung cancer mortality and low serum lycopene levels is more complex and multi-directional than suggested. As a result, more research is needed to investigate the physiological mechanisms underlying the link between lycopene and a reduction in lung cancer mortality.
Ethical Approval
The NHANES protocol was reviewed and approved by the Institutional Review Board (IRB) of the National Center of Health Statistics. This current lung cancer study was exempt from ethics review by the Georgia Southern University IRB.
Author Contributions:
JZ: Study design and analysis; EAG, PS, GS, SC, AAY, NA: Analyzed, interpreted the data, and drafted the manuscript; All authors: Manuscript revision.
Acknowledgments:
The NHANES has been developed and funded by multiple federal agencies including the National Center for Health Statistics. The fieldwork was conducted by Westat, Inc. under contract by the National Center for Health Statistics. Also, the authors would like to express our appreciation to Dr. Sewuese Akuse, Ms. Paige Perry, and Ms. Morgan Fleming for editing the manuscript for accuracy and consistency.
References
1. Naz A, Butt MS, Sultan MT, Qayyum MM, .Niaz RS. Watermelon lycopene and allied health claims. EXCLI J. 2014, 13:650-60. eCollection
2. Borel P, Desmarchelier C, Dumont U, Halimi C, Lairon D, Page D, Sebedio JL, Buisson C, Buffiere C, & Remond D. Dietary calcium impairs tomato lycopene bioavailability in healthy humans. British Journal of Nutrition. 2016 Dec;116(12):2091-2096.
3. Woodside JV, McGrath AJ, Lyner N, & McKinley MC. Carotenoids and health in older people. Maturitas, 2015, 80(1), 63-68.
4. Cristina Trejo-Solís, Jose Pedraza-Chaverrí, Mónica Torres-Ramos, Dolores Jiménez-Farfán, Arturo Cruz Salgado, Norma Serrano-García, Laura Osorio-Rico, Julio Sotelo, “Multiple Molecular and Cellular Mechanisms of Action of Lycopene in Cancer Inhibition“, Evidence-Based Complementary and Alternative Medicine, vol. 2013, Article ID 705121, 17 pages, 2013. https://doi.org/10.1155/2013/705121
5. Anese M, Mirolo G, Fabbro A, Lippe G. Lycopene bioaccessibility and bioavailability from processed foods. J Sci Ind Res, 2013; 72, 543-547.
6. Moran NE, Erdman JW, & Clinton SK. Complex interactions between dietary and genetic factors impact lycopene metabolism and distribution. Archives of Biochemistry and Biophysics, 539(2), 10.1016/j.abb.2013.06.017. http://doi.org/10.1016/j.abb.2013.06.017
7. Qiu X, Yuan Y, Vaishnav A, Tessel MA, Nonn L, & van Breemen RB. Effects of lycopene on protein expression in human primary prostatic epithelial cells. Cancer Prev Res, 2013; 6(5), 419-427.
8. Singla N, Jain S, Kachroo M, Gakhar A. Review on Functions of Dietary Agents and Plants in Cancer Prevention and Treatment. Syst Rev Pharm 2013;4:31-9.
9. Ford ES, Li C, Cunningham TJ, & Croft JB. Associations between antioxidants and all-cause mortality among US adults with obstructive lung function. British Journal of Nutrition, 2014, 112(10): 1662-1673.
10. Chaiyasit W, McClements DJ, & Decker EA. The relationship between the physicochemical properties of antioxidants and their ability to inhibit lipid oxidation in bulk oil and oil-in-water emulsions. Journal of Agricultural and Food Chemistry, 53(12), 4982-4988.
11. Gajowik A, & Dobrzynska MM. Lycopene-antioxidant with radioprotective and anticancer properties. A review. Roczniki Państwowego Zakładu Higieny, 2014, 65(4).
12. Havermann S, Büchter C, Koch K, & Wätjen W. Role of Oxidative Stress in the Process of Carcinogenesis. In Studies on Experimental Toxicology and Pharmacology (pp. 173-198). Springer International Publishing. 2015
13. Ono M, Takeshima M, Nakano S. Mechanism of the Anticancer Effect of Lycopene (Tetraterpenoids). Enzymes. 2015;37:139-66. doi: 10.1016/bs.enz.2015.06.002. Epub 2015 Jul 21.
14. Wang Y, Jacobs EJ, Newton CC, & McCullough ML. Lycopene, tomato products, and prostate cancer‐specific mortality among men diagnosed with nonmetastatic prostate cancer in the Cancer Prevention Study II Nutrition Cohort. International Journal of Cancer, 2016, 138(12), 2846-2855.
15. Martí R, Roselló S, Cebolla-Cornejo J. Tomato as a Source of Carotenoids and Polyphenols Targeted to Cancer Prevention. Cancers (Basel). 2016 Jun 20;8(6). pii: E58. doi: 10.3390/cancers8060058.
16. Centers for Disease Control and Prevention. Lung cancer: What Are the Risk Factors for Lung Cancer? (2021). Available at https://www.cdc.gov/cancer/lung/basic_info/risk_factors.htm
17. Valavanidis A, Vlachogianni T, Fiotakis K, & Loridas S. Pulmonary Oxidative Stress, Inflammation and Cancer: Respirable Particulate Matter, Fibrous Dusts and Ozone as Major Causes of Lung Carcinogenesis through Reactive Oxygen Species Mechanisms. Int. J. Environ. Res. Public Health 2013, 10(9), 3886-3907; doi:3390/ijerph10093886
18. Csordas A, Bernhard D. The biology behind the atherothrombotic effects of cigarette smoke. Nature Reviews Cardiology 2013, vol 10: 219–230 doi:10.1038/nrcardio.2013.8.
19. Palozza P, Simone RE, Catalano A, & Mele MC. Tomato lycopene and lung cancer prevention: from experimental to human studies.Cancers, 2011, 3(2), 2333-2357.
20. Cuzick J, Thorat MA, Andriole G, Brawley OW, Brown PH, Culig Z, Eeles RA, Ford LG, Hamdy FC, Holmberg L, Ilic D, Key TJ, La Vecchia C, Lilja H, Marberger M, Meyskens FL, Minasian LM, Parker C, Parnes HL, Perner S, Rittenhouse H, Schalken J, Schmid HP, Schmitz-Dräger BJ, Schröder FH, Stenzl A, Tombal B, Wilt TJ, Wolk A. Prevention and Early Detection of Prostate Cancer. Lancet Oncol. 2014 October ; 15(11): e484–e492. doi:10.1016/S1470-2045(14)70211-6.
21. Kotecha R, Takami A, Espinoza JL. Dietary phytochemicals and cancer chemoprevention: a review of the clinical evidence. Oncotarget. 2016, 7(32):52517-52529. doi: 10.18632/oncotarget.9593.
22. Mezzomo N & Ferreira SRS. Carotenoids Functionality, Sources, and Processing by Supercritical Technology: A Review. Journal of Chemistry, 2016, Volume 2016. http://dx.doi.org/10.1155/2016/3164312.
23. Centers for Disease Control and Prevention/National Center for Health Statistics (NCHS). NCHS Data Linkage: 2017. https://www.cdc.gov/nchs/data/datalinkage/LinkageTable.pdf
24. Centers for Disease Control and Prevention/National Center for Health Statistics (NCHS). NCHS Data Linkage: 2021. https://www.cdc.gov/nchs/data/datalinkage/LinkageTable.pdf
25. Twum F, Morte N, Wei Y, Nkemjika S, Liu F, & Zhang J. Red blood cell folate and cardiovascular deaths among hypertensive adults, an 18-year follow-up of a national cohort. Hypertension Research, 2020; 43, 938–947
26. Giovannucci E. Tomatoes, tomato-based products, lycopene, and cancer: a review of the epidemiologic literature. J Natl Cancer Inst. 1998; 91(4):317-31
27. Ito Y, Wakai K, Suzuki K, Ozasa K, Watanabe Y, Seki N, Ando M, Nishino Y, Kondo T, Ohno Y, & Tamakoshi A. Lung cancer mortality and serum levels of carotenoids, retinol, tocopherols, and folic acid in men and women: a case-control study nested in the JACC Study. Journal of Epidemiology, 2005, 15(Supplement_II): S140-S149.
28. Lagiou P, BenetouV, Tebelis N, Papas AA, Naska A, & Trichopoulou Plasma carotenoid levels in relation to tobacco smoking and demographic factors. Int J Vitam Nutr Res. 2003; 73(3):226-31.
29. Stavrides JC. Lung carcinogenesis: pivotal role of metals in tobacco smoke. Free Radical Biology and Medicine, 2006, 41(7): 1017-1030.
30. Suwannalert P, Boonsiri P, Khampitak T, Khampitak K, Sriboonlue P, & Yongvanit P. The levels of lycopene, alpha]-tocopherol and a marker of oxidative stress in healthy northeast Thai elderly. Asia Pacific Journal of Clinical Nutrition, 2007, 16, 27-30.
31. Liu C, Russell RM, & Wang XD. Lycopene supplementation prevents smoke-induced changes in p53, p53 phosphorylation, cell proliferation, and apoptosis in the gastric mucosa of ferrets. J Nutr, 2006, 136(1): 106-111
![]()